Search

Search










trans-Latanoprost is a critical geometric isomer impurity of the glaucoma drug Latanoprost, featuring a trans-double bond configuration. Primarily used as a certified reference standard, it is essential for pharmaceutical quality control, method validation, and stability testing to ensure final drug purity. Key selling points include exceptional chemical purity (>98%) with fully confirmed stereochemistry, supported by comprehensive characterization data (NMR, MS) for regulatory filings. Our product complies with ISO 17034 guidelines, providing traceable Certificates of Analysis crucial for FDA and EMA audits. Ideal for generic manufacturers proving bioequivalence, it enables precise quantification of impurities to meet strict safety limits. Packaged in light-resistant vials to prevent degradation, we guarantee stability and reliability. Sold strictly as a laboratory reference material for analytical use only; not for human or animal administration.
Items | Specifications | Results |
Appearance | Colorless to yellowish viscous oil liquid | Conforms |
Identification | HPLC: Sample and standard exhibits same retention time | Conforms |
Water | ≤0.5% | 0.20% |
Loss on drying | ≤1.0% | 0.31% |
Residual on solvents | Ethanol:≤0.5% | 0.30% |
Total impurities | ≤5.0% | 3.90% |
Purity | ≥95.0% | 96.1% |
Conclusion | The product conforms to the Enterprise Standard. | |
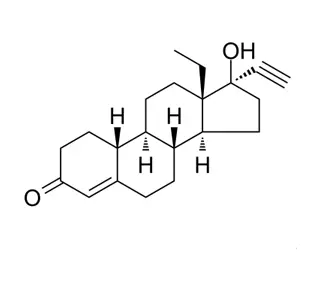
trans-Latanoprost (also known as Latanoprost Impurity A or (5E,15S)-Latanoprost, CAS No. 1235141-39-5) is a specific geometric isomer and impurity of the active pharmaceutical ingredient (API) Latanoprost.
Chemical Nature: While commercial Latanoprost contains a cis double bond at the 5,6-position of the heptenoate chain, trans-Latanoprost features a trans (E) configuration at this position. It also typically refers to the 15S stereoisomer.
Primary Use: It is not a therapeutic drug itself. Instead, it serves as a critical reference standard and impurity marker in the pharmaceutical industry.
Quality Control (QC): Used to validate HPLC/GC methods to ensure the final Latanoprost drug product (used for glaucoma treatment) meets strict purity limits (usually <0.1% or <0.5% for this impurity).
Stability Studies: Used to monitor the degradation of Latanoprost formulations over time, as the cis-to-trans isomerization can occur under stress conditions (light, heat).
Process Development: Helps chemists optimize synthesis routes to minimize the formation of this unwanted isomer.
Key Selling Points
Targeted at pharmaceutical manufacturers, QC laboratories, and regulatory affairs departments, the selling points include:
High Purity & Characterization:
Supplied with high chemical purity (>95% or >98%) and confirmed stereochemistry. Essential for accurate calibration and reliable impurity quantification.
Comprehensive characterization data provided (NMR, MS, IR, HPLC) to support regulatory filings (IND/NDA/ANDA).
Regulatory Compliance:
Manufactured and documented in accordance with ISO 17034 (Reference Material Producer) guidelines.
Includes a detailed Certificate of Analysis (COA) traceable to international standards, crucial for FDA, EMA, and NMPA audits.
Critical for Safety & Efficacy Validation:
Enables manufacturers to prove their product is free from excessive levels of this specific geometric isomer, ensuring patient safety and drug efficacy.
Stability & Packaging:
Packaged in light-resistant, temperature-controlled vials to prevent isomerization during storage, guaranteeing the integrity of the reference standard.
Global Supply for Generic Drug Approval:
Essential for generic drug companies seeking approval; having access to certified impurity standards is a mandatory requirement for demonstrating bioequivalence and quality parity with the brand-name drug (Xalatan).
Important Notice: This product is sold strictly as a chemical reference standard or research material for laboratory quality control and analytical purposes only. It is not for human or animal administration, nor for use as a finished pharmaceutical product.




Fortunachem Provides Not Only Professional Chemical Products But Also Professional Help
Keeping you up-to-date with all the latest information, news, and events about Fortunachem!

Quick Links
Add:
E-mail:
 English
English  Español
Español  français
français  العربية
العربية  中文
中文